PROTOCOL FOR INSTITUTIONAL REVIEW BOARD (IRB)
1. Protocol Submission and Review
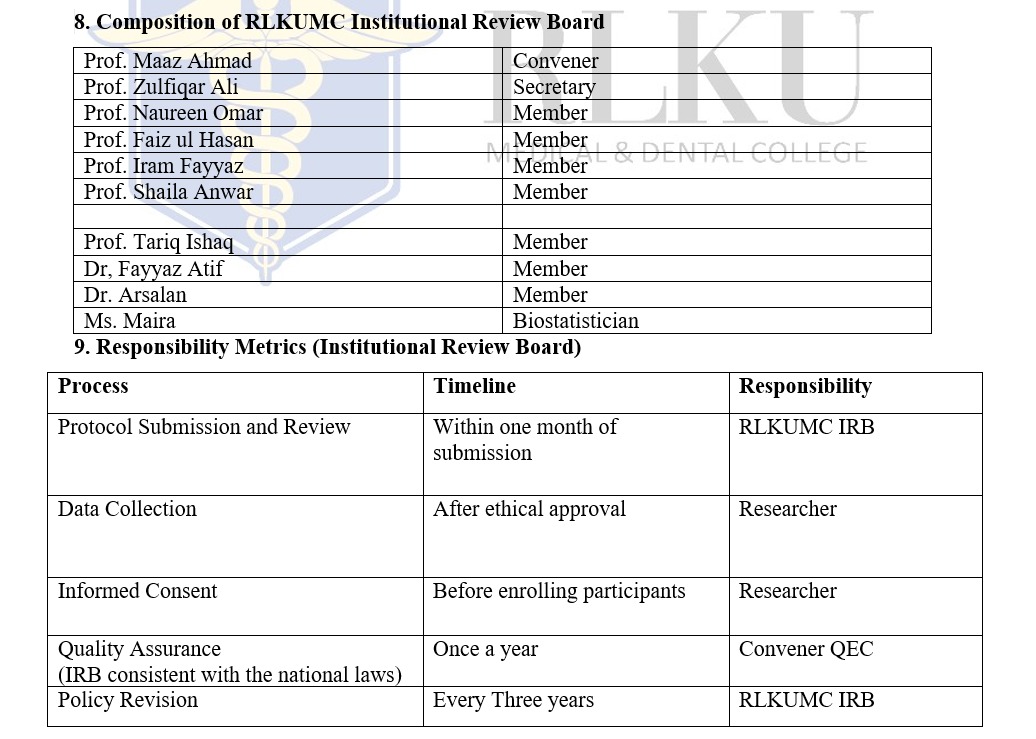
1.1 Researchers shall submit research protocols to the IRB through the IRB Application form.1.2 The IRB shall review protocols within a month of submission, considering the level of risk involved.
1.3 The IRB shall conduct initial reviews for all protocols to ensure compliance with ethical standards, regulatory requirements, and the IRB policy.
1.4 Expedited review procedures may be used for minimal risk studies as determined by the IRB Chairperson.
2. Conflict of Interest Management
2.1 IRB members shall disclose any potential conflicts of interest related to the research under review.2.2 The IRB Chairperson or designated official shall assess the disclosed conflicts of interest and determine the appropriate course of action.
2.3 Members with conflicts of interest may be excluded from reviewing or voting on the research protocol in question.
3. Informed Consent Process
3.1 Researchers shall develop an informed consent process consistent with ethical standards and regulatory requirements provided in IRB application form.3.2 The IRB shall review and approve the informed consent documents before participant recruitment.
3.3 The IRB shall ensure the informed consent process includes adequate information about risks, benefits, alternatives, and participant rights.
3.4 In cases where waivers or alterations of informed consent are requested, the IRB shall review and make determinations based on regulatory criteria.
4. Communication and Conflict Resolution
4.1 The IRB shall promptly and fairly address and resolve conflicts or disagreements arising during the review process promptly and fairly.4.2 Communication channels, such as email or designated meetings, shall be used to facilitate timely and efficient communication.
6. Quality Assurance
6.1 The RLKUMC IRB policy is consistent with all applicable national laws, regulations, and RLKUMC policies governing the protection of human research subjects.7. Policy Review and Revision
7.1 The IRB policy shall be reviewed and revised at-least every three years to reflect changes in laws, regulations, and ethical standards.7.2 Proposed revisions to the IRB policy shall undergo review and approval by the appropriate institutional authorities.
7.3 Updated versions of the IRB policy shall be communicated to all relevant stakeholders.